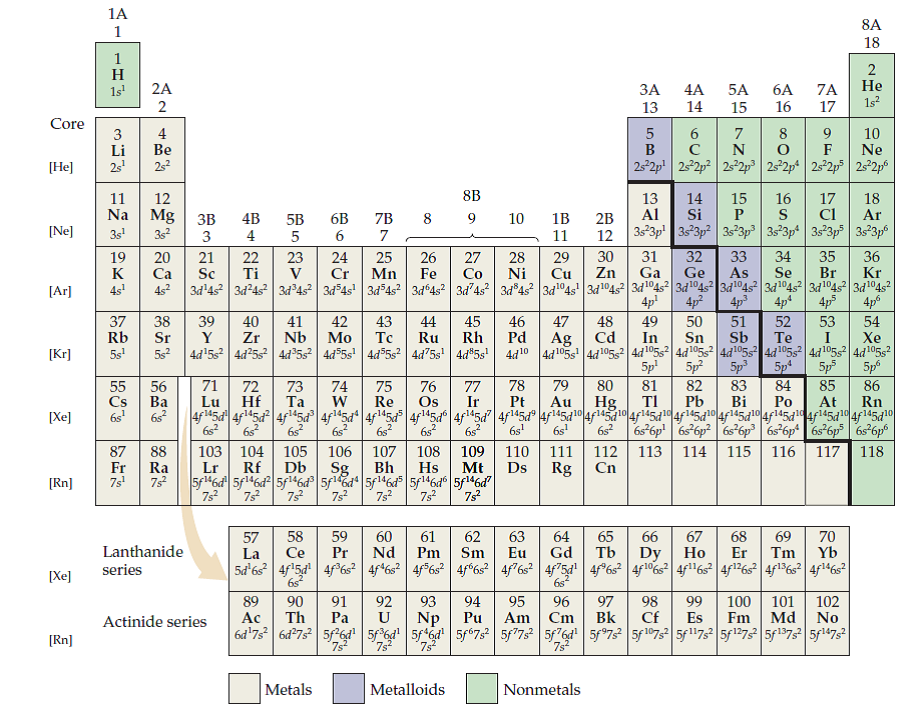
If, for example, according to Higgins, the force between the ultimate particle of oxygen and the ultimate particle of nitrogen were 6, then the strength of the force would be divided accordingly, and likewise for the other combinations of ultimate particles (see illustration). In 1789, William Higgins published views on what he called combinations of "ultimate" particles, which foreshadowed the concept of valency bonds. The quest for the underlying causes of valence led to the modern theories of chemical bonding, including the cubical atom (1902), Lewis structures (1916), valence bond theory (1927), molecular orbitals (1928), valence shell electron pair repulsion theory (1958), and all of the advanced methods of quantum chemistry. 
The concept of valence was developed in the second half of the 19th century and helped successfully explain the molecular structure of inorganic and organic compounds. William Higgins' combinations of ultimate particles (1789) The etymology of the words valence (plural valences) and valency (plural valencies) traces back to 1425, meaning "extract, preparation", from Latin valentia "strength, capacity", from the earlier valor "worth, value", and the chemical meaning referring to the "combining power of an element" is recorded from 1884, from German Valenz. This definition differs from the IUPAC definition as an element can be said to have more than one valence. Valence is defined by the IUPAC as: The maximum number of univalent atoms (originally hydrogen or chlorine atoms) that may combine with an atom of the element under consideration, or with a fragment, or for which an atom of this element can be substituted.Īn alternative modern description is: The number of hydrogen atoms that can combine with an element in a binary hydride or twice the number of oxygen atoms combining with an element in its oxide or oxides. The two tables below show examples of different compounds, their structural formulas, and the valences for each element of the compound. The structural formula of a compound represents the connectivity of the atoms, with lines drawn between two atoms to represent bonds. Phosphorus has a valence 3 in phosphine ( PH 3) and a valence of 5 in phosphorus pentachloride ( PCl 5), which shows that an element may exhibit more than one valence. Chlorine, as it has a valence of one, can be substituted for hydrogen in many compounds. In methane, carbon has a valence of 4 in ammonia, nitrogen has a valence of 3 in water, oxygen has a valence of 2 and in hydrogen chloride, chlorine has a valence of 1. The valence is the combining capacity of an atom of a given element, determined by the number of hydrogen atoms that it combines with. Valence is not to be confused with the related concepts of the coordination number, the oxidation state, or the number of valence electrons for a given atom. In most compounds, the valence of hydrogen is 1, of oxygen is 2, of nitrogen is 3, and of carbon is 4. Double bonds are considered to be two bonds, triple bonds to be three, quadruple bonds to be four, quintuple bonds to be five and sextuple bonds to be six. Valence is generally understood to be the number of chemical bonds that each atom of a given chemical element typically forms. In chemistry, the valence (US spelling) or valency (British spelling) of an atom is a measure of its combining capacity with other atoms when it forms chemical compounds or molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
